Emerging technologies in cancer detection

Exciting, modern technologies for cancer detection are under development in academic and industrial laboratories worldwide. This chapter provides a synopsis of technologies reaching greater importance as they advance toward clinical practice. These methods include significant advances in current methods as well as fundamentally new platforms. We place a special emphasis on point-of-care technologies for use in clinical settings as well as novel methods for use as at-home measurements and wearable devices. We also provide a synopsis on the involvement of artificial intelligence-based data analytics such as machine learning algorithms in both existing and developing assessments.
Nature Nanotechnology: A wavelength-induced frequency filtering method for fluorescent nanosensors in vivo

Fluorescent nanosensors hold the potential to revolutionize life sciences and medicine. However, their adaptation and translation into the in vivo environment is fundamentally hampered by unfavourable tissue scattering and intrinsic autofluorescence. Here we develop wavelength-induced frequency filtering (WIFF) whereby the fluorescence excitation wavelength is modulated across the absorption peak of a nanosensor, allowing the emission signal to be separated from the autofluorescence background, increasing the desired signal relative to noise, and internally referencing it to protect against artefacts. Using highly scattering phantom tissues, an SKH1-E mouse model and other complex tissue types, we show that WIFF improves the nanosensor signal-to-noise ratio across the visible and near-infrared spectra up to 52-fold. This improvement enables the ability to track fluorescent carbon nanotube sensor responses to riboflavin, ascorbic acid, hydrogen peroxide and a chemotherapeutic drug metabolite for depths up to 5.5 ± 0.1 cm when excited at 730 nm and emitting between 1,100 and 1,300 nm, even allowing the monitoring of riboflavin diffusion in thick tissue. As an application, nanosensors aided by WIFF detect the chemotherapeutic activity of temozolomide transcranially at 2.4 ± 0.1 cm through the porcine brain without the use of fibre optic or cranial window insertion. The ability of nanosensors to monitor previously inaccessible in vivo environments will be important for life-sciences research, therapeutics and medical diagnostics.
Nature Digital Medicine: Grass-roots entrepreneurship complements traditional top-down innovation in lung and breast cancer

The majority of biomedical research is funded by public, governmental, and philanthropic grants. These initiatives often shape the avenues and scope of research across disease areas. However, the prioritization of disease-specific funding is not always reflective of the health and social burden of each disease. We identify a prioritization disparity between lung and breast cancers, whereby lung cancer contributes to a substantially higher socioeconomic cost on society yet receives significantly less funding than breast cancer. Using search engine results and natural language processing (NLP) of Twitter tweets, we show that this disparity correlates with enhanced public awareness and positive sentiment for breast cancer. Interestingly, disease-specific venture activity does not correlate with funding or public opinion. We use outcomes from recent early-stage innovation events focused on lung cancer to highlight the complementary mechanism by which bottom-up “grass-roots” initiatives can identify and tackle under-prioritized conditions.
wePool.AI

wePool AI provides a computational testing strategy that leverages Artificial Intelligence to predict a subject’s probability of testing positive for COVID-19, and uses it to segment test populations into distinct pools.
Role: Advisor
Temporal Imaging of Live Cells by High-Speed Confocal Raman Microscopy

Label-free live cell imaging was performed using a custom-built high-speed confocal Raman microscopy system. For various cell types, cell-intrinsic Raman bands were monitored. The high-resolution temporal Raman images clearly delineated the intracellular distribution of biologically important molecules such as protein, lipid, and DNA. Furthermore, optical phase delay measured using quantitative phase microscopy shows similarity with the image reconstructed from the protein Raman peak. This reported work demonstrates that Raman imaging is a powerful label-free technique for studying various biomedical problems in vitro with minimal sample preparation and external perturbation to the cellular system.
Transcutaneous Measurement of Essential Vitamins Using Near-Infrared Fluorescent Single-Walled Carbon Nanotube Sensors

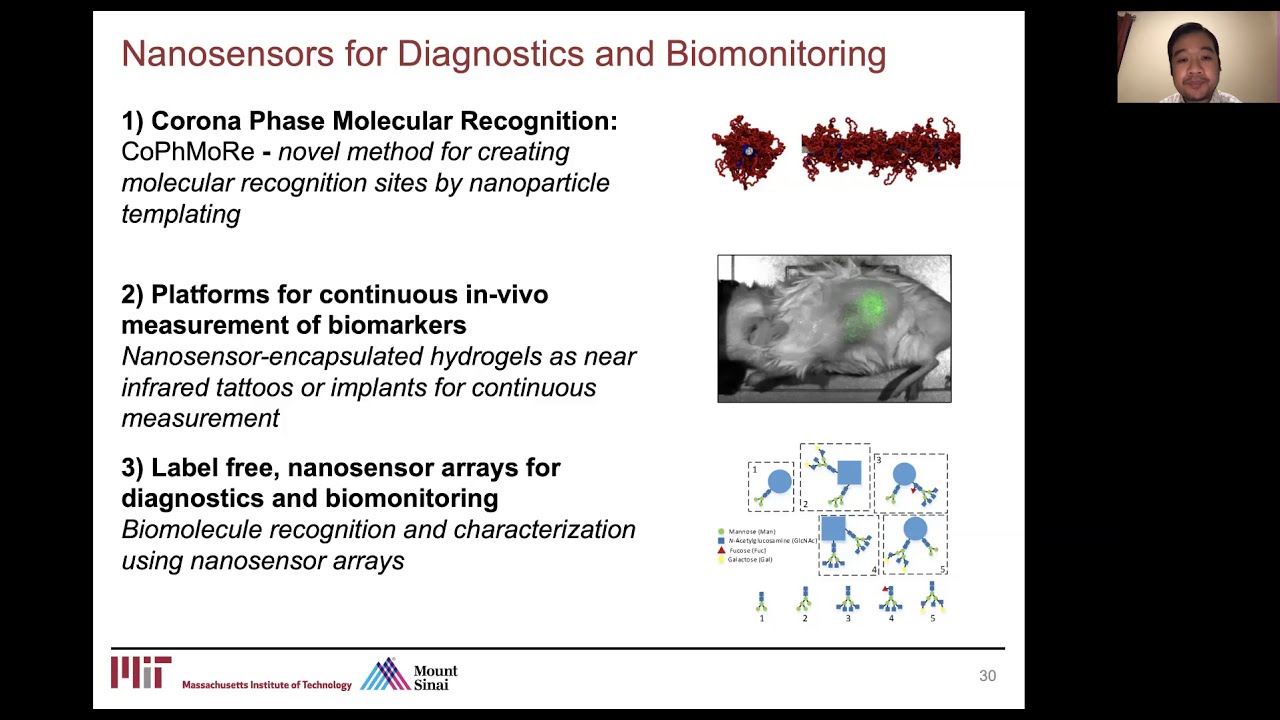
Vitamins such as riboflavin and ascorbic acid are frequently utilized in a range of biomedical applications as drug delivery targets, fluidic tracers, and pharmaceutical excipients. Sensing these biochemicals in the human body has the potential to significantly advance medical research and clinical applications. In this work, a nanosensor platform consisting of single-walled carbon nanotubes (SWCNTs) with nanoparticle corona phases engineered to allow for the selective molecular recognition of ascorbic acid and riboflavin, is developed. The study provides a methodological framework for the implementation of colloidal SWCNT nanosensors in an intraperitoneal SKH1-E murine model by addressing complications arising from tissue absorption and scattering, mechanical perturbations, as well as sensor diffusion and interactions with the biological environment. Nanosensors are encapsulated in a polyethylene glycol diacrylate hydrogel and a diffusion model is utilized to validate analyte transport and sensor responses to local concentrations at the boundary. Results are found to be reproducible and stable after exposure to 10% mouse serum even after three days of in vivo implantation. A geometrical encoding scheme is used to reference sensor pairs, correcting for in vivo optical and mechanical artifacts, resulting in an order of magnitude improvement of p-value from 0.084 to 0.003 during analyte sensing.
Hackers Without Borders | AI LA Life Summit 2020

Hackers Without Borders | AI LA Life Summit 2020 Hackers Without Borders | AI LA Life Summit 2020
2020 Beckman Symposium: From biochemical nanosensors to imaging to informatics to COVID-19 convalescent plasma – developing diagnostics and therapies for clinical medicine
2017 Arnold O. Beckman Postdoctoral Fellow Freddy Nguyen, MD, PhD from MIT, presents his research at the 2020 Beckman Symposium.
Transfusion reactions associated with COVID-19 convalescent plasma therapy for SARS-CoV-2

Background: Convalescent plasma (CP) for treatment of severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) has shown preliminary signs of effectiveness in moderate to severely ill patients in reducing mortality. While studies have demonstrated a low risk of serious adverse events, the comprehensive incidence and nature of the spectrum of transfusion reactions to CP is unknown. We retrospectively examined 427 adult inpatient CP transfusions to determine incidence and types of reactions, as well as clinical parameters and risk factors associated with transfusion reactions. Study Design and Methods: Retrospective analysis was performed for 427 transfusions to 215 adult patients with coronavirus 2019 (COVID‐19) within the Mount Sinai Health System, through the US Food and Drug Administration emergency investigational new drug and the Mayo Clinic Expanded Access Protocol to Convalescent Plasma approval pathways. Transfusions were blindly evaluated by two reviewers and adjudicated by a third reviewer in discordant cases. Patient demographics and clinical and laboratory parameters were compared and analyzed. Results: Fifty‐five reactions from 427 transfusions were identified (12.9% incidence), and 13 were attributed to transfusion (3.1% incidence). Reactions were classified as underlying COVID‐19 (76%), febrile nonhemolytic (10.9%), transfusion‐associated circulatory overload (9.1%), and allergic (1.8%) and hypotensive (1.8%) reactions. Statistical analysis identified increased transfusion reaction risk for ABO blood group B or Sequential Organ Failure Assessment scores of 12 to 13, and decreased risk within the age group of 80 to 89 years. Conclusion: Our findings support the use of CP as a safe, therapeutic option from a transfusion reaction perspective, in the setting of COVID‐19. Further studies are needed to confirm the clinical significance of ABO group B, age, and predisposing disease severity in the incidence of transfusion reaction events.
Neutralizing Antibody Responses in COVID-19 Convalescent Sera

Passive transfer of antibodies from COVID-19 convalescent patients is being used as an experimental treatment for eligible patients with SARS-CoV-2 infections. The United States Food and Drug Administration’s (FDA) guidelines for convalescent plasma initially recommended target antibody titers of 160. We evaluated SARS-CoV-2 neutralizing antibodies in sera from recovered COVID-19 patients using plaque reduction neutralization tests (PRNT) at moderate (PRNT50) and high (PRNT90) stringency thresholds. We found that neutralizing activity significantly increased with time post symptom onset (PSO), reaching a peak at 31–35 days PSO. At this point, the number of sera having neutralizing titers of at least 160 was approximately 93% (PRNT50) and approximately 54% (PRNT90). Sera with high SARS-CoV-2 antibody levels (>960 enzyme-linked immunosorbent assay titers) showed maximal activity, but not all high-titer sera contained neutralizing antibody at FDA recommended levels, particularly at high stringency. These results underscore the value of serum characterization for neutralization activity.
Implantable Nanosensors for Human Steroid Hormone Sensing In Vivo Using a Self-Templating Corona Phase Molecular Recognition

Dynamic measurements of steroid hormones in vivo are critical, but steroid sensing is currently limited by the availability of specific molecular recognition elements due to the chemical similarity of these hormones. In this work, a new, self‐templating synthetic approach is applied using corona phase molecular recognition (CoPhMoRe) targeting the steroid family of molecules to produce near infrared fluorescent, implantable sensors. A key limitation of CoPhMoRe has been its reliance on library generation for sensor screening. This problem is addressed with a self‐templating strategy of polymer design, using the examples of progesterone and cortisol sensing based on a styrene and acrylic acid copolymer library augmented with an acrylated steroid. The pendant steroid attached to the corona backbone is shown to self‐template the phase, providing a unique CoPhMoRE design strategy with high efficacy. The resulting sensors exhibit excellent stability and reversibility upon repeated analyte cycling. It is shown that molecular recognition using such constructs is viable even in vivo after sensor implantation into a murine model by employing a poly (ethylene glycol) diacrylate (PEGDA) hydrogel and porous cellulose interface to limit nonspecific absorption. The results demonstrate that CoPhMoRe templating is sufficiently robust to enable a new class of continuous, in vivo biosensors.
Nature Medicine: Convalescent plasma treatment of severe COVID-19: a propensity score–matched control study

Coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is a new human disease with few effective treatments. Convalescent plasma, donated by persons who have recovered from COVID-19, is the acellular component of blood that contains antibodies, including those that specifically recognize SARS-CoV-2. These antibodies, when transfused into patients infected with SARS-CoV-2, are thought to exert an antiviral effect, suppressing virus replication before patients have mounted their own humoral immune responses. Virus-specific antibodies from recovered persons are often the first available therapy for an emerging infectious disease, a stopgap treatment while new antivirals and vaccines are being developed. This retrospective, propensity score–matched case–control study assessed the effectiveness of convalescent plasma therapy in 39 patients with severe or life-threatening COVID-19 at The Mount Sinai Hospital in New York City. Oxygen requirements on day 14 after transfusion worsened in 17.9% of plasma recipients versus 28.2% of propensity score–matched controls who were hospitalized with COVID-19 (adjusted odds ratio (OR), 0.86; 95% confidence interval (CI), 0.75–0.98; chi-square test P value = 0.025). Survival also improved in plasma recipients (adjusted hazard ratio (HR), 0.34; 95% CI, 0.13–0.89; chi-square test P = 0.027). Convalescent plasma is potentially effective against COVID-19, but adequately powered, randomized controlled trials are needed.
